  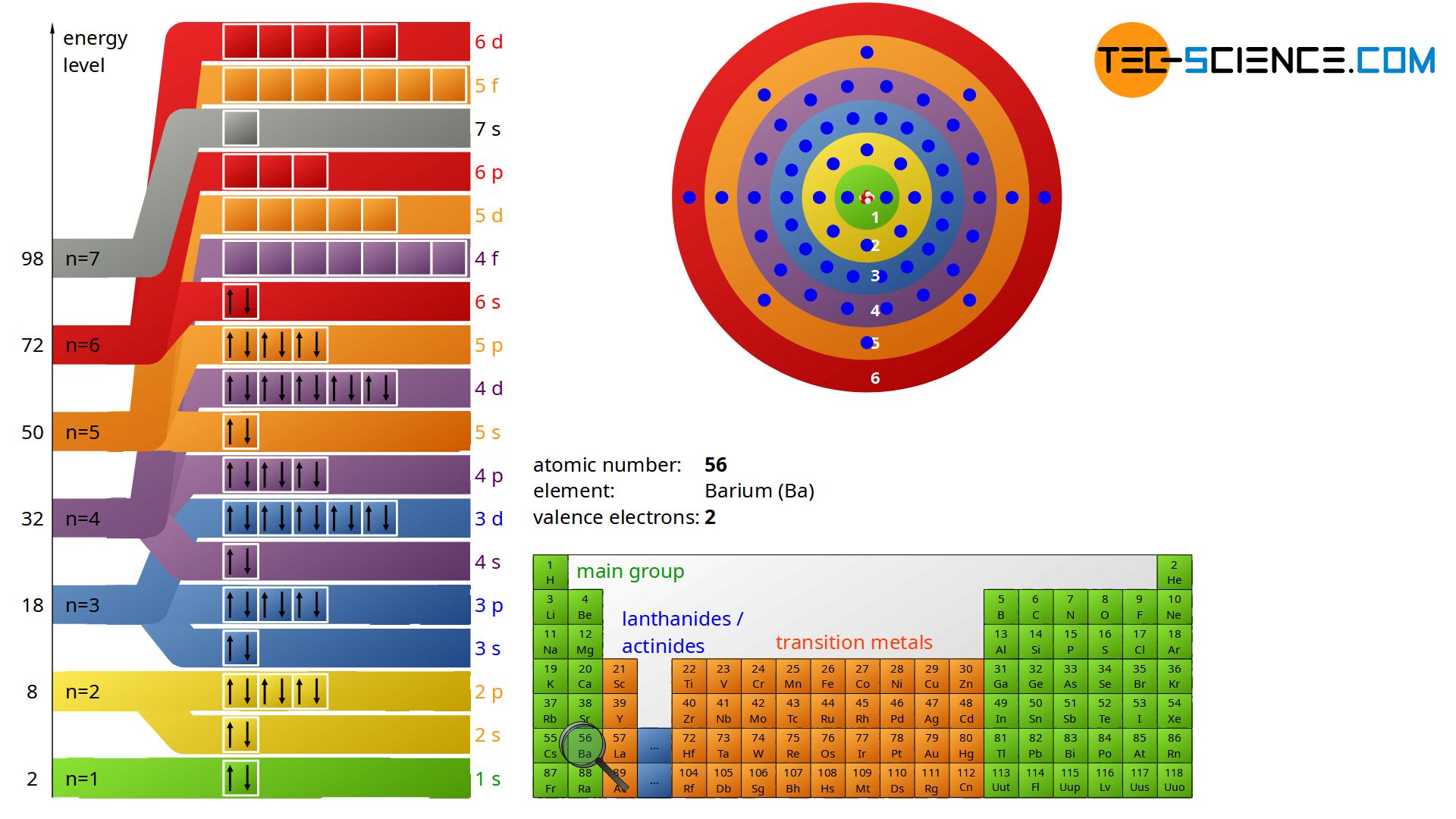 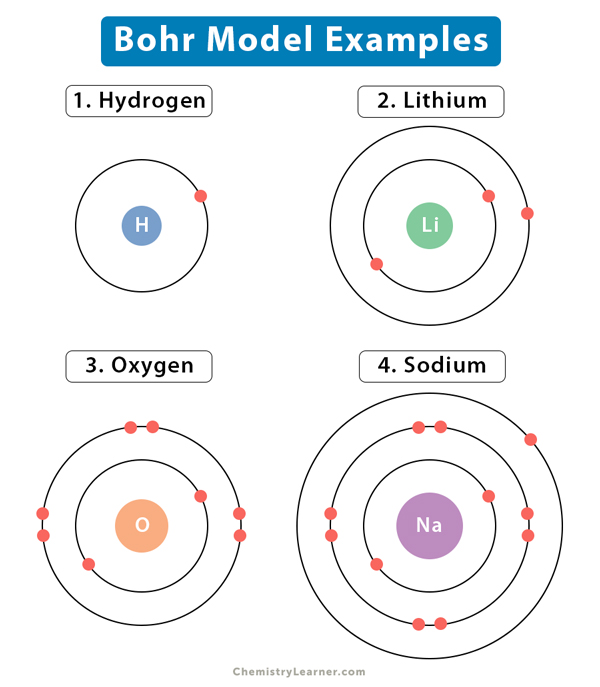
He concluded that electron will have more energy if it is located away from the nucleus whereas electrons will have less energy if it located near the nucleus. E two is equal to negative 3.4, and E three is equal to negative 1.51 electron volts. Hence, the Lewis structure of oxygen can be drawn as follows: Properties of Oxygen. According to the Bohr Atomic model, a small positively charged nucleus is surrounded by revolving negatively charged electrons in fixed orbits. The energy for the first energy level is equal to negative 13.6. (It was a running joke that any theory of atomic and molecular spectra could be destroyed by throwing a book of data at it, so complex were the spectra. The Bohr model of the oxygen atom shows that oxygen contains 6 electrons in its valence shell. Since Bohr’s model involved only a single electron, it could also be applied to the single electron ions He +, Li 2 +, Be 3 +, and so forth, which differ from hydrogen only in their nuclear charges, and so one-electron atoms and ions are collectively referred to as hydrogen-like atoms. And i found a counterexample : Plus, in all my classcial mechanics classes, the centripetal force was inward. In my opinion, the centripetal force should be directed towards the center, and the electrostatic force should be directed outward. But, in spite of years of efforts by many great minds, no one had a workable theory. Its the Bohr Model for electron moving around the nucleus. Neil Bohr, one of the founders of quantum mechanics, was interested in the much-debated topic of the time the structure of the atom.Numerous atomic models, including the theory postulated by J.J Thompson and the discovery of the nucleus by Ernest Rutherford, had emerged. Maxwell and others had realized that there must be a connection between the spectrum of an atom and its structure, something like the resonant frequencies of musical instruments. Quantum mechanics emerged in the mid-1920s. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
